- View Mobile Number
iirtdelhi@gmail.com
Chemistry Services
Leading Manufacturers, Exporters, Wholesaler, Retailer of 5 Batch Analysis Service, Accelerated Storage Stability Testing Service, Agro Chemicals Testing Services, Analytical Method Development Service, Characterization and Identification Testing Services, Chemical Equivalence Service, Cold Storage Stability Testing Chamber, Cold Storage Stability Testing Chamber, Laboratory Testing Services, Long Term Accelerated Storage Stability Testing Service, Method Validation Service, Pesticide Residue Analysis Service, Physico-Chemical Studies, Storage Stability Testing Service and Testing OF Agrochemicals and Industrial Chemicals from Ghaziabad.
Five-batch analysis is necessary for identifying and quantifying impurities within the production batches of substances to obtain the minimum regulatory closure of 98% w/w required for successful registration. For over 20 years, Charles River chemists have developed expertise in the separation, identification, synthesis and quantification of impurities that may be present as a very small proportion of structurally similar or isometric materials.
5 Batch Analysis refers to the comprehensive testing and evaluation of five separate production batches of a product to ensure consistency, quality, and compliance withspecifications. It is a critical part of product validation and quality assurance in manufacturing.
Long-term storage stability testing is a critical process used to assess how a product’s quality, safety, and efficacy change over an extended period when stored under recommended conditions. This testing helps determine the product’s shelf life and supports the establishment of a scientifically justified expiration date.
By simulating real-time storage environments—such as controlled temperature, humidity, and light exposure—this testing evaluates changes in physical appearance, chemical composition, potency, and microbial stability over time. It ensures that the product remains effective and safe for use throughout its intended lifespan.
Long-term stability data are essential for regulatory submissions, product labeling, and ensuring consistent performance during distribution and storage. This testing is particularly important for pharmaceuticals, medical devices, and biologics, where degradation could affect therapeutic outcomes or patient safety.
Agrochemicals and industrial chemicals are broad classes of substances extensively used in agriculture and various industrial applications. Agrochemicals include products like pesticides, herbicides, and fertilizers that enhance crop yield and protect plants from pests and diseases. Industrial chemicals encompass a wide range of compounds used in manufacturing, construction, textiles, and other industrial processes.
Testing and evaluation of these chemicals are essential to ensure their effectiveness, human and environmental safety, and regulatory compliance. Comprehensive studies are conducted to assess their toxicity, residue levels, persistence in the environment, and potential impact on human health, animals, soil, and water systems.
Regulatory authorities require thorough documentation and safety data before these chemicals can be approved for commercial use. Rigorous evaluation helps minimize risks, promotes responsible usage, and ensures that their benefits do not come at the cost of long-term ecological or health hazards.
Analytical Method Development is the process of designing, refining, and validating a procedure to accurately, precisely, and consistently detect, identify, and quantify specific analytes within a given sample. This process is fundamental in various industries, including pharmaceuticals, chemicals, environmental testing, and food safety, where reliable analytical results are critical for product quality, regulatory compliance, and safety assurance.
The method development process involves selecting appropriate analytical techniques (such as HPLC, GC, UV-Vis, or spectroscopy), optimizing parameters like solvent systems, detection wavelengths, and flow rates, and ensuring the method is robust, reproducible, and suitable for its intended purpose.
Once developed, the method undergoes validation to confirm performance characteristics such as accuracy, precision, specificity, linearity, limit of detection (LOD), and limit of quantification (LOQ). A well-developed analytical method ensures confidence in the results and supports informed decision-making throughout product development and quality control.
Identification and characterization are essential analytical processes used to confirm the identity and determine the physical, chemical, and structural properties of a substance or material. These procedures play a critical role in research and development, quality control, and regulatory compliance across various industries, including pharmaceuticals, medical devices, and chemicals.
Identification ensures that the correct substance is being used or studied, verifying its authenticity and consistency. Characterization, on the other hand, provides detailed information about the material’s attributes, such as composition, molecular structure, purity, and stability.
Together, these processes help ensure product safety, efficacy, and quality by detecting impurities, verifying raw materials, and supporting formulation decisions. They are vital at every stage of a product 's lifecycle—from initial development through to manufacturing and post-market surveillance—ensuring that materials meet defined specifications and regulatory standards.
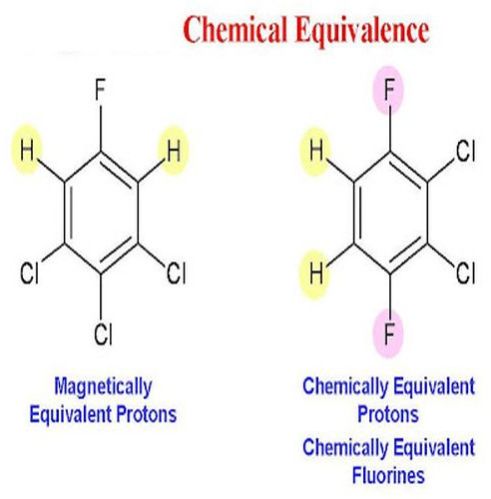
Chemical equivalence refers to the evaluation and confirmation that two or more chemical substances or formulations possess the same chemical identity, composition, and potency. This assessment ensures that the substances are effectively interchangeable for their intended application without compromising safety, quality, or performance.
Chemical equivalence is particularly important in the development of generic drugs, medical devices, and pharmaceutical formulations, where it must be demonstrated that the active ingredient in a test product matches that of the reference product. This includes verification of molecular structure, concentration, and purity levels.
Establishing chemical equivalence supports regulatory approvals, aids in quality control, and helps maintain therapeutic consistency across different products. It also ensures patients receive the same clinical benefits, regardless of the product source. Analytical methods such as spectroscopy, chromatography, and potency assays are commonly used to confirm chemical equivalence in both research and manufacturing settings.
| Business Type | Exporter, Supplier |
| Application | Evaluate Stability And Integrity Of Products Stored At Low Temperatures |
| Type | Testing Equipment |
| Material | Various |
| Temperature Range | Refrigerated (2-8°C) Or Frozen Conditions (-20°C Or Below) |
| Quality Assurance | Ensures Product Maintains Quality, Safety, And Efficacy During Cold Storage |
Cold Storage Stability Testing evaluates the stability and integrity of products stored at lowtemperatures, typically refrigerated (2–8°C) or frozen conditions (-20°C or below). This test ensures that the product maintains its quality, safety, and efficacy during cold storage.
Food process manufacturing involves the conversion of raw agricultural materials into food products. Encompassing several production phases, many of which require strict temperature control for the preservation of ingredients, food processing is a manufacturing route for transitioning intermediate products into finished goods.
Refrigerators, freezers, temperature-controlled chambers and cold rooms are employed throughout the entirety of the food manufacturing process, creating ideal environments for product preservation and long-term storage during and after the manufacturing process.
Cold Storage Stability Testing evaluates the stability and integrity of products stored at lowtemperatures, typically refrigerated (2–8°C) or frozen conditions (-20°C or below). This test ensures that the product maintains its quality, safety, and efficacy during cold storage.
Food process manufacturing involves the conversion of raw agricultural materials into food products. Encompassing several production phases, many of which require strict temperature control for the preservation of ingredients, food processing is a manufacturing route for transitioning intermediate products into finished goods.
Refrigerators, freezers, temperature-controlled chambers and cold rooms are employed throughout the entirety of the food manufacturing process, creating ideal environments for product preservation and long-term storage during and after the manufacturing process.
Laboratory Testing Services involve a broad spectrum of analytical and bioassay evaluations performed under controlled laboratory conditions to assess the safety, efficacy, quality, and environmental impact of various substances, materials, and products. These services play a crucial role in supporting research and development, regulatory compliance, and quality assurance across multiple industries, including pharmaceuticals, chemicals, medical devices, environmental science, and consumer goods.
Testing may include chemical analysis, microbiological testing, toxicity studies, stability testing, and performance evaluations, depending on the product and its intended use. By generating accurate and reliable data, laboratory testing ensures products meet industry standards, regulatory requirements, and consumer safety expectations.
These services are essential for identifying potential risks, improving product design, and facilitating market approval, ultimately helping companies maintain high standards of safety and quality throughout the product lifecycle.
Long Term Storage Stability Testing evaluates how the quality, safety, and efficacy of a product change over an extended period under recommended storage conditions. It determines the product’s shelf life and expiration date.
Method validation is a crucial process in analytical and testing laboratories to ensure that a specific test method is reliable, reproducible, and appropriate for its intended use. It involves a systematic assessment to confirm that the method consistently produces accurate, precise, and consistent results under defined experimental conditions.
This process is essential for demonstrating that a method can effectively measure the target analyte in a given sample matrix, whether for quality control, regulatory compliance, or research purposes. Method validation typically evaluates key performance characteristics such as accuracy, precision, specificity, sensitivity, linearity, range, and robustness.
By validating a method, laboratories can ensure confidence in their test results, support data integrity, and comply with regulatory requirements set by agencies like the FDA, EMA, or ISO. Ultimately, method validation safeguards the quality and reliability of analytical data used in pharmaceuticals, medical devices, food safety, and environmental testing.
Residue analysis is a critical process used to detect and quantify trace levels of chemical substances—such as pesticides, herbicides, veterinary drugs, and other contaminants—in agricultural products. These products include fruits, vegetables, grains, and animal-derived foods like meat, milk, and eggs. The primary goal of residue analysis is to ensure that these food items meet established safety standards and comply with regulatory limits set by national and international authorities.
Advanced analytical techniques such as GC-MS (Gas Chromatography-Mass Spectrometry) and LC-MS/MS (Liquid Chromatography-Tandem Mass Spectrometry) are commonly used to detect even minute amounts of residues with high accuracy.
This testing plays a vital role in protecting consumer health, supporting international trade, and maintaining public trust in food safety. Regular residue analysis also helps monitor the proper use of agrochemicals and veterinary medicines in food production systems.
Physico-chemical studies involve the detailed analysis of a substance’s physical and chemical properties, providing crucial data for understanding its behavior, performance, and safety. These studies are essential in evaluating attributes such as appearance, pH, solubility, melting point, viscosity, chemical composition, and impurities.
They play a key role in quality control, ensuring consistency and compliance with product specifications throughout manufacturing. Additionally, physico-chemical studies support formulation development by helping select appropriate ingredients and optimize product stability and performance.
These evaluations are also vital for stability testing, determining how a product changes over time under various environmental conditions, and identifying factors that may affect its shelf life. Furthermore, physico-chemical data are often required for regulatory submissions, helping demonstrate product safety, efficacy, and compatibility with other materials. Overall, these studies form the foundation for the safe and effective development of pharmaceuticals, medical devices, and other health-related products.
Cold Storage Stability Testing is performed to assess the stability, quality, and integrity of products stored under low-temperature conditions. These conditions typically include refrigerated storage (2–8°C) and frozen storage (-20°C or below), depending on the product’s requirements.
This testing ensures that a product retains its safety, efficacy, and physical characteristics—such as appearance, potency, and chemical composition—throughout its intended shelf life while stored in cold environments. It is especially critical for temperature-sensitive products like biologics, vaccines, and certain pharmaceuticals, which may degrade or lose effectiveness if exposed to improper conditions.
The evaluation involves storing samples under defined cold storage settings for a specified duration and conducting periodic tests to monitor changes. Cold Storage Stability Testing plays a vital role in regulatory compliance, supply chain management, and product labeling, ensuring that the product remains effective and safe when stored and transported under recommended cold conditions.
Agrochemicals and Industrial Chemicals are broad categories of substances widely used in agriculture and various industries, respectively. Testing and evaluation of these chemicals are crucial to ensure their efficacy, safety, environmental impact, and regulatory compliance.